|
3/4/2024 0 Comments En iso 13485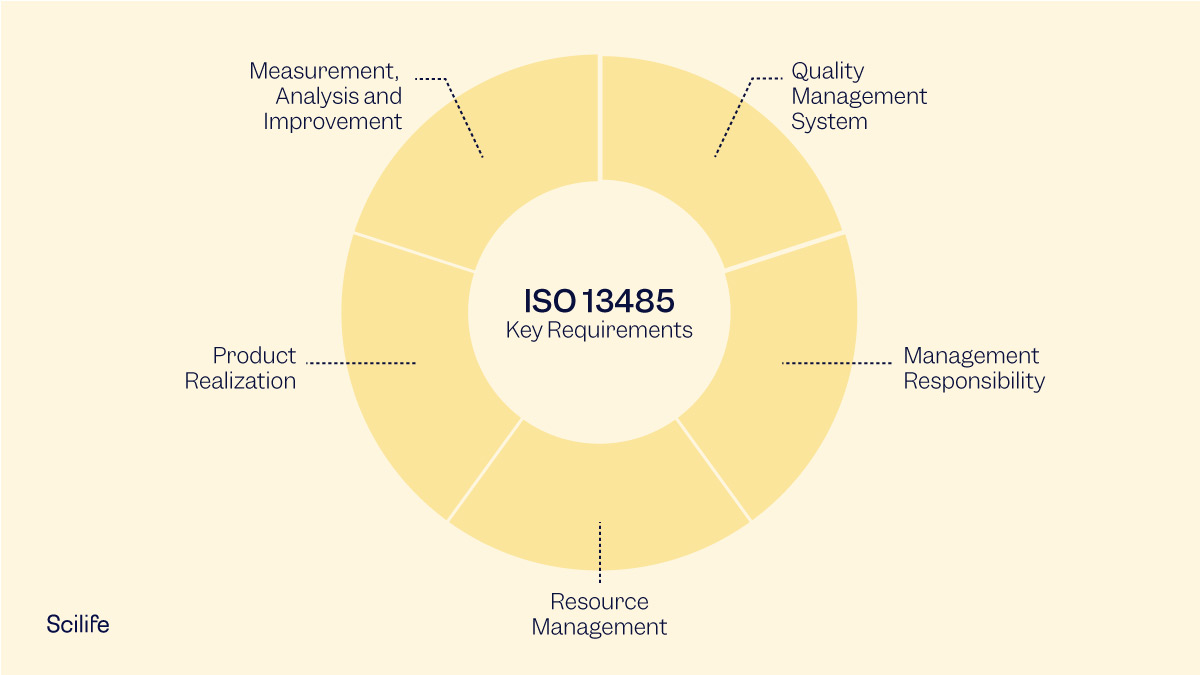 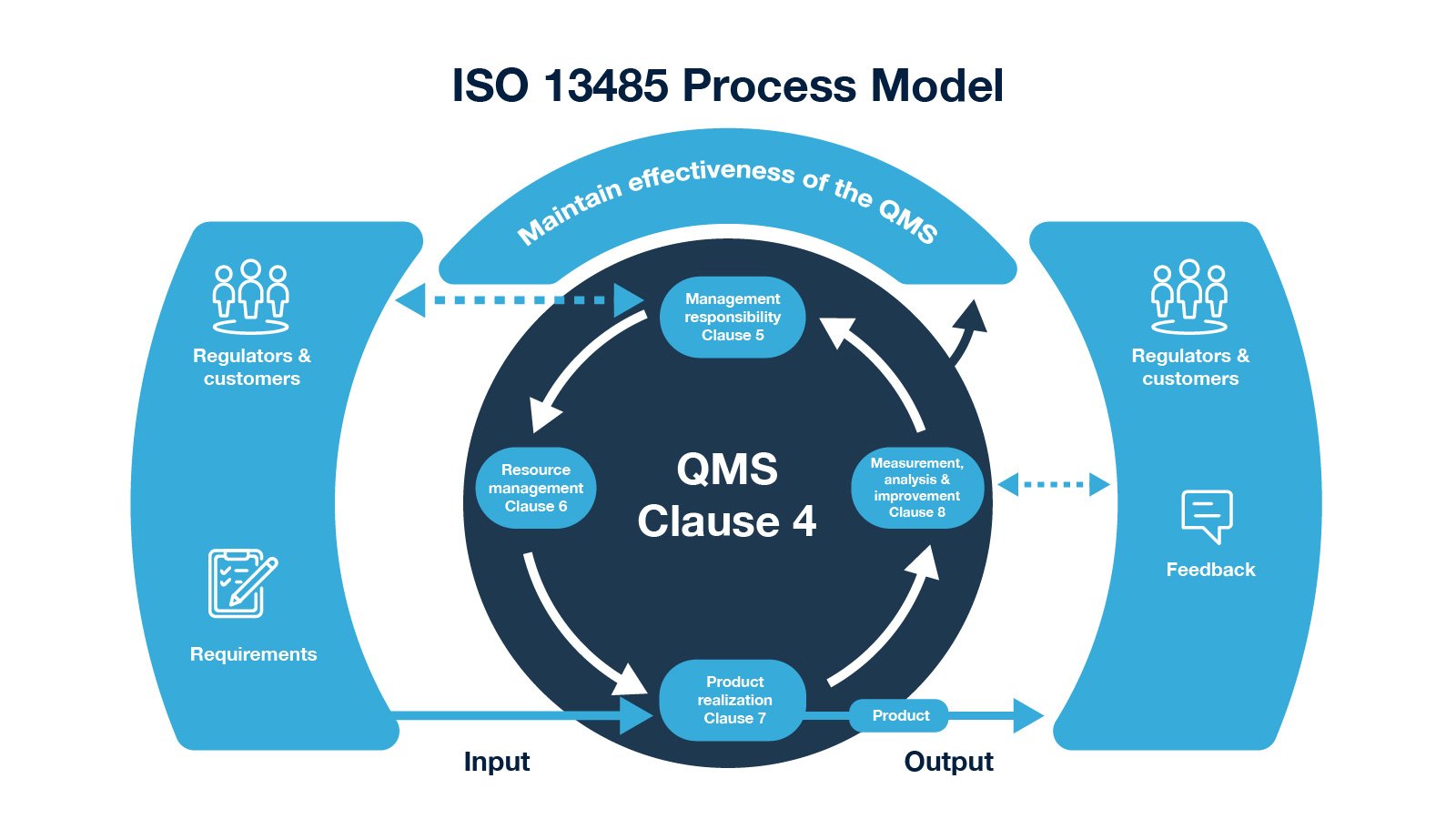
Requirements of ISO 13485 are applicable to organizations regardless of their size and regardless of their type except where explicitly stated. The supplier or external party can voluntarily choose to conform to the requirements of the standard or can be required by contract to conform. ISO 13485 can also be used by suppliers or external parties that provide products, including quality management system-related services. ISO 13485 can be used by organizations that are involved in one or more stages of the life-cycle, including design and development, production, storage and distribution, installation, or servicing of a medical device and design and development or provision of associated activities (e.g. It can be used to demonstrate the ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements. The adoption of a quality management system is a strategic decision of an organization. ISO 13485 is an international standard that specifies the requirements of a quality management system for medical devices.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
